Posts Tagged: biofuel
The food vs. fuel debate: Growing biofuel in the U.S.
However, estimates suggest that growing crops to produce that much biofuel would require 40 to 50 million acres of land, an area roughly equivalent in size to the entire state of Nebraska.
“If we convert cropland that now produces food into fuel production, what will that do to our food supply?” asks Maggi Kelly, UC Cooperative Extension specialist and the director of the UC Agriculture and Natural Resources Statewide IGIS Program. “If we begin growing fuel crops on land that isn't currently in agriculture, will that come at the expense of wildlife habitat and open space, clean water and scenic views?”
Kelly and UC Berkeley graduate student Sarah Lewis are conducting research to better understand land-use options for growing biofuel feed stock. They used a literature search, in which the results of multiple projects conducted around the world are reviewed, aggregated and compared.
“When food vs. fuel land questions are raised in the literature, authors often suggest fuel crops be planted on ‘marginal land,'” Kelly said. “But what does that actually mean? Delving into the literature, we found there was no standard definition of ‘marginal land.'”
Kelly and Lewis' literature review focused on projects that used geospatial technology to explicitly map marginal, abandoned or degraded lands specifically for the purpose of planting bioenergy crops. They narrowed their search to 21 papers from 2008 to 2013, and among them they found no common working definition of marginal land.
“We have to be careful when we talk about what is marginal. We have to be explicit about our definitions, mapping and modeling,” Kelly said. “In our lab, we are trying to understand the landscape under multiple lenses and prioritize different uses and determine how management regimes impact the land.”
The research report, titled Mapping the Potential for Biofuel Production on Marginal Lands: Differences in Definitions, Data and Models across Scales, was published in the International Journal for Geo-Information.
Click here for this story in Spanish.
An initiative to improve energy security and green technologies is part of UC Agriculture and Naturalist Resources Strategic Vision 2025.
Giant King Grass harvested at UC research center
The first crop of Giant King Grass, a hybrid grass with biofuel potential, was harvested at the UC Desert Research and Extension Center in Holtville this month, reported Ethanol Producer Magazine.
Giant King Grass is a fast-growing, high-yield grass that grows under a variety of soil conditions, according to Viaspace Green Energy Inc. It is propagated vegetatively and, with sufficient rain or irrigation, can grow 15 to 18 feet high in six months.
At the UC Desert Rec, scientists compared two planting processes:
- Planting single nodes that grow into individual plants with some space between them.
- Planting whole stalks continuously end to end, which results in a dense row of plants about six inches apart.
Preliminary results showed the whole stalk planting germinated earlier and grew more quickly. The individual plants had a significant number of skips where the nodes failed to germinate.
Two harvesting regimens were tested:
- Harvest when the plant is 6 to 8 feet tall every two months for animal feed and to produce biogas for anaerobic digestion.
- Harvest when the plant is 15 to 18 feet tall for bioenergy applications, such as direct combustion in a power plant, energy pellets or cellulosic biofuels.
"It was 108 degrees when I arrived in Holtville last Monday evening (Sept. 8, 2014) at 6 p.m.," said Carl Kukkonen, CEO of Viaspace. "Giant King Grass is planted in the worst soil at the University of California site, and still the results are good. I am pleased that Giant King Grass grows well in this extremely hot and dry environment."
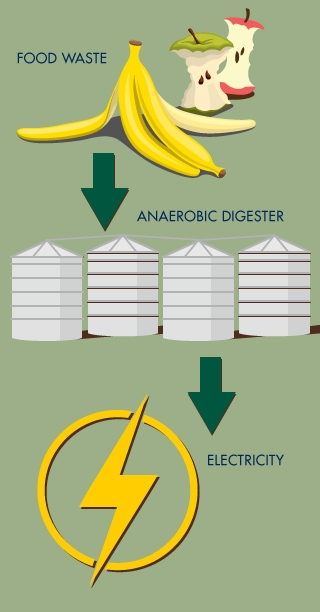
Biodigester turns campus waste into campus energy
Last week, on Earth Day, the university and Sacramento-based technology partner CleanWorld unveiled the UC Davis Renewable Energy Anaerobic Digester (READ) at the campus' former landfill. Here, the anaerobic digestion technology Zhang invented is being used inside large, white, oxygen-deprived tanks. Bacterial microbes in the tanks feast on campus and community food and yard waste, converting it into clean energy that feeds the campus electrical grid.
“This technology can change the way we manage our solid waste,” Zhang said. “It will allow us to be more economically and environmentally sustainable."
It is the third commercial biodigester CleanWorld has opened using Zhang's technology within the past two years and is the nation's largest anaerobic biodigester on a college campus.
The system is designed to convert 50 tons of organic waste to 12,000 kWh of renewable electricity each day using state-of-the-art generators, diverting 20,000 tons of waste from local landfills each year. It is expected to reduce greenhouse gas emissions by 13,500 tons per year.
The READ BioDigester encompasses several of the university's goals: reducing campus waste in a way that makes both economic and environmental sense, generating renewable energy, and transferring technology developed at UC Davis to the commercial marketplace.
The biodigester will enable the more than 100 million tons of organic waste each year that is currently being landfilled in the U.S. to be converted to clean energy and soil products. The READ BioDigester is a closed loop system, moving from farm to fork to fuel and back to farm. Whatever is not turned into biogas to generate renewable electricity can be used as fertilizer and soil amendments — 4 million gallons of it per year, which could provide natural fertilizers for an estimated 145 acres of farmlands each day.
Nearly half of the organic waste, or feedstock, needed to operate the biodigester to full benefit will come from UC Davis dining halls, animal facilities and grounds. CleanWorld is working with area food processing and distribution centers to supply the remaining amount. Meanwhile, UC Davis will earn 100 percent of the project's green energy and carbon credits and receive all of the electricity generated.
Anaerobic digestion is an age-old process. However, Zhang's patented technology made it more efficient — capable of eating a broader variety and bigger quantity of waste, turning it into clean energy faster and more consistently than other commercial anaerobic biodigesters.
View a video about the UC David biodigester here:
(This blog post is condensed from a UC Davis news release about the biodigester.)
Additional information:
- Read the full press release
- Download biodigester photos
- Vine video: From lunch to lights
- Visit http://www.cleanworld.com/
Homegrown/Backyard Oyster Mushroom Cultivation
I recently attended a 4-hour workshop on homegrown/backyard mushroom cultivation organized by Biofuel Oasis in Berkeley, California. Patty and Ray Lanier of Mushroom Maestros located in both Oakland and in Lake County, California, were the featured speakers of this seminar.
The workshop began with a Powerpoint presentation on a brief history and folklore of the mushroom, the life cycle of a mushroom, and how mushrooms play a part in our ecological system. After the Powerpoint presentation, Patty and Ray led the group through some hands-on projects, which is the focus of this blog entry.
I was surprised to learn how easy it was/is to cultivate some varieties of mushrooms at home, specifically, the oyster mushroom and wine cap mushroom (king stropharia). Below are instructions on how to cultivate oyster mushrooms at home. Cultivating wine cap mushrooms is even easier and will be discussed in one of my upcoming blog entries.
Oyster Mushroom
Oyster mushrooms are aggressive and eager to grow.
Materials needed:
1. Large pot (e.g., a canning pot)
2. Large colander
3. Large mesh bag (e.g, an onion bag from the supermarket)
4. 1 bag of mushroom innoculent (can be bought on-line)
5. 1 cylindrical plastic bag (e.g., the plastic bags that newspapers come in or a large plastic bread bag)
6. 1 bag/jar of mushroom grain spawn (can be purchased easily on-line)
Step 1: Pasteurize (not sterilize) clean wheat straw (note: not hay, but straw) which you can purchase inexpensively from a well-stocked feed store, in 150 - 180 F degree heat for 45 - 60 minutes. It may help to place the wheat straw in a mesh bag like an onion bag, to keep the straw together.
Step 2: After you have pasteurized the straw, let it dry by placing it in a colander.
Step 3: Sterilize all work surfaces, tools, your hands, etc., with rubbing alcohol. Empty the mesh bag of its straw contents and spread the straw (straw should be warm to the touch) onto a sterilized surface so that so that the straw layer is 2-3 inches thick.
Step 4: Take handfuls of the mushroom grain spawn and toss it onto the straw until the grains dot the surface of the straw. Work the grain spawn into the straw.
Step 5: Stuff the straw into your cylindrical plastic bag, compressing the straw enough so that the bag is firmly (but not overly) packed. Tie the bag off.
Step 6: Take a nail or a sharp pair of scissors and pierce the bag, making random 1-inch cuts all over the bag, but not to the point where the straw begins to fall out. This is to aerate the bag while keeping a moist environment for the spawn. Wipe the bag down with alcohol to keep it sterile.
Step 7: Place the bag in a shady area indoors (i.e., does not receive direct sunlight) in an area where the temperature is kept between 60 – 75 degrees. Keep the bag moist but not damp, by spraying with a spray bottle filled with water. You should see signs of oyster mushrooms in 2 weeks (the log will turn white), at which time, you can either keep the bag indoors or locate it in a shady part of your yard, protected from the wind and sun and heat. When the caps of mushrooms flatten out, it is time to harvest. Don't wait too long, as the mushrooms can grow past its prime in just 1 day.
For more information on either Biofuel Oasis or Mushroom Maestros, please see www.biofueloasis.com and http://www.mushroommaestros.com/, respectively.

Pasteurizing straw. (photos by Betty Homer)

Mushroom logs hung outside after being indoors for 2 weeks.

Oyster mushrooms peeping out.

Oyster mushrooms ready to harvest.
Biofuel research may keep tobacco industry from going up in smoke
The troubled tobacco industry may be getting some good news for a change. UC scientists are engineering the tobacco plant to produce oils that, when extracted, can serve as drop-in biofuels to power airplanes, cars and other machines.
Research success would allow farmers who have been growing tobacco for generations to continue the tradition for a different purpose, while taking advantage of an infrastructure established to serve the diminishing cigarette, cigar and snuff markets.
Peggy G. Lemaux, UC Cooperative Extension specialist, and Anastasios Melis and Krishna Niyogi, Agricultural Experiment Station faculty in the Department of Plant and Microbial Biology at UC Berkeley, are lead researchers in the project.
“There are several reasons we are modifying tobacco to produce biofuel,” Lemaux said, “It is a high biomass crop. If you want to extract oil, then the more biomass you have, the more oil you get. And, since tobacco is not a food source, tobacco production for biofuel would not have an impact on global food markets or find its way into the food supply. Finally, tobacco farmers are anxious to produce a product that is more acceptable to the public."
However, there are no tobacco varieties that produce oil at high levels. Scientists know that certain algae do turn sunlight energy into oil, and that sparked an idea.
A research consortium that includes Lemaux, Melis and Niyogi and researchers at the Lawrence Berkeley National Laboratory and the University of Kentucky is taking genes, primarily from algae, that produce oil and inserting them into tobacco plants to make oils in leaves.
To do that, the consortium scientists are using very small pieces of tobacco leaves to introduce the desired algal genes, where they become a heritable trait of the tobacco plant. The small pieces of leaf tissue are then placed in a Petri dish, the tissues that contain the introduced genes identified, and then the tissues grow into a new plant, every cell of which contains the algal genes.
“We hope the new plant will make the same kind of oils that algae do,” Lemaux said. “We can then use organic solvents to extract the oils out of the leaves.”
Much more research is required before tobacco plants that produce biofuel at commercial levels will be available to growers, but Lemaux is encouraged by the preliminary results.
The research is funded with a three-year $4.8 million grant from a U.S. Department of Energy, Advanced Research Projects Agency – Energy program termed PETRO, Plants Engineered to Replace Oil. ARPA-E funds high-risk, high-reward research projects to find potential alternatives to fossil fuels.
See a video about this research below: