Posts Tagged: fertilizer
Skip landscape fertilization during the drought
"When plants are under drought stress, we don't want to promote a lot of leafy growth," says Missy Gable in the fifth installment of UC ANR's six-part video series on saving water in the landscape. "If using fertilizer, choose a fertilizer low in nitrogen, or don't fertilize this year."
The UC Master Gardener Program provides a detailed description of landscape fertilizer needs on its California Backyard Orchard website. Visit the website to learn about the various nutrient needs of growing trees and shrubs, symptoms of nutrient deficiencies, and how much fertilizer should generally be applied each year.
View the latest video here:
Additional videos in the UC ANR video series on saving water in the landscape.
Early-morning watering is best
Remove weeds from your landscape
The videos are also available on the UC ANR YouTube channel.
Coming next week, a video about using fertilizers under drought conditions.
An initiative to improve California water quality, quantity and security is part of the UC Division of Agriculture and Natural Resources Strategic Vision 2025.
Author: Jeannette Warnert
Drought-focused soil nutrient management series offered for farmers online
Beginning in November, a free, drought-focused soil nutrient management series for farmers will be hosted online by the University of California Sustainable Agriculture Research & Education Program (UC SAREP), FarmsReach and Sustainable Conservation.
“Farmers and ranchers have to continually adapt their management of soil nutrients to changing conditions,” said Aubrey White, UC SAREP's communication coordinator. “Adaptation during this extreme drought presents a new challenge for growers and researchers alike. This forum dedicated to the issues farmers will face next season is an opportunity to share resources, research and ideas for success.”
Kicking off on Nov. 17, the Nutrient Management Solutions series will offer the agriculture community:
- Online presentations, videos or Q&A with farmers and UC Cooperative Extension advisors on nutrient management and soil fertility, with special focus on tree crops, grapes and dairy farms.
- Facilitated online discussions in the active FarmsReach Conversations, moderated by Series presenters. (Join the Nutrient Management Solutions Group in FarmsReach to participate.)
- A new “Soil Nutrient Management Toolkit” in FarmsReach, with selected practical resources and info sheets for farmers of all crop and product types.
The online series is part of the Solution Center for Nutrient Management—a growing resource for nutrient management research and information, online and in-person created by UC SAREP.
The presentations, videos and facilitated online Q&A will be hosted in three sections:
- Nov. 17-30 – Nutrient Management in Times of Drought: Tree Crops
- December (dates to be announced) – Nutrient Management in Times of Drought: Wine Grapes
- January (dates to be announced) – Nutrient Management in Times of Drought: Dairy Forage Crops
To get updates and announcements, or to share your ideas for the drought-focused Nutrient Management Solutions series, sign up for free at www.farmsreach.com. You can also go directly to the online group within FarmsReach, http://www.farmsreach.com/nutrient-mgmt-series, and follow news on Twitter at #AgSolutionCenter.
About UC SAREP
The University of California Sustainable Agriculture Research & Education Program (UC SAREP) a program in UC Division of Agriculture and Natural Resources, provides leadership and support for scientific research and education in agricultural and food systems that are economically viable, conserve natural resources and biodiversity, and enhance the quality of life in the state's communities. SAREP serves farmers, farmworkers, ranchers, researchers, educators, regulators, policy makers, industry professionals, consumers and community organizations across the state.
About FarmsReach
Founded in 2007, FarmsReach is a network that connects small- and medium-scale farms to the products, support and services they need to be successful. By partnering with farmer members and agriculture organizations, FarmsReach offers a growing suite of services that empower farmers to make better business decisions, access new markets, preserve the environment and strengthen rural communities.
About Sustainable Conservation
Sustainable Conservation partners with business, agriculture and government to find practical ways that the private sector can protect clean air, clean water and healthy ecosystems. The independent nonprofit organization leads powerful collaborations that produce lasting solutions and sustain the vitality of both the economy and the environment.
Now I Know My N-P-Ks (Next time won't you plant with me?) Part Three
We have come to the third and final installment of my series discussing the three macronutrients essential for a healthy garden. My first blog was about nitrogen, why it is needed in the garden, what it might look like if there is a deficiency, and what do do about correcting it. The second article was about phosphorus. This final submission of the series is about potassium.
To catch you up, these three essential macronutrients are contained in the bag or bottle of fertilizer you purchase. However, unless you are informed, you will simply see three meaningless numbers listed there, e.g. 5-10-5; or 12-10-5. You must know that these numbers refer to the percentage of each macronutrient included, and these are always in a specific order; nitrogen (N), phosphorus (P), and potassium (K). In the first of these examples, the fertilizer contains 5% nitrogen, 10% phosphorus, and 5 % potassium. Use these numbers to decide which product you need to purchase for your gardening needs.
So why do we need potassium? Potassium helps regulate photosynthesis, moisture content of plant cells and stomata, which controls the carbon dioxide exchange. Potassium helps move vital nutrients around in the plant itself. It also aides in the formation of proteins, which directly effect the nutritional value of the fruit. Lastly, potassium aids the soil as it is responsible for fixing nitrogen in legumes which need a potassium rich soil.
What would a potassium deficiency look like? It would be much subtler than a nitrogen or phosphorus deficiency, even to the extent that you might not notice it. Your plants may appear a bit smaller or thinner than expected, but they do not look "sick". There are a few symptoms to be on the lookout for, however. If your plants appear weak and spindly, attract more pests than usual, and/or plants bear small, thin-skinned fruits that are lacking in flavor, you most likely have a potassium deficiency. This deficiency, as with the other two, can be remedied by composting.
Another highly recommended remedy is the use of greensand. This is really green, and has the consistency of sand. Add 10 pounds per 100 square feet to a new garden. Add it to your compost pile also to ensure against future. It also contains iron, magnesium, calcium, phosphorus, and more than 30 trace elements.
Wood ash contains about 5% potassium in a relative quick release form. Spread on a new garden without plants (late fall or winter), as the ash can actually burn plants. A third recommendation is granite dust. It will provide a water solvable potassium that your plants can use immediately. Granite dust is about 3% immediately available, and 3% available over time. It will not effect your pH balance. (pH balance? Ah, fodder for another blog!)
Ground kelp has been noted as a source of potassium, but its percentage is less than the above mentioned additives.
In reviewing these macronutrients, it is clear that all of them are essential. Most likely, like the vitamins our bodies need, if your garden is lacking in one, it is lacking in others. As I review the differences in the appearance of the deficiencies, I see more similarities the differences. I have come to the conclusion that I must not only keep feverishly composting, but actually increase the amount I compost, as compost contains the nutrients required by most plants. Then, if that fails, and deficiencies continue to appear, I will have my soil professionally analyzed. There are testing kits available for around $11.00, but in researching these, I found numerous reports of unreliability. Instead, I would recommending using the UC Davis Analytical Lab. Information on how to use this resource, the costs, and the procedures are available if you simply put UC Davis Analytical Lab in your search engine.
I have learned a lot from writing a three segmented blog about N-P-K, but I must admit what has really increased is my ever growing awe of nature and how it comes together and works to produce what we need. Little, tiny, invisible nutrients effecting big leafy, greens--with or without my understanding. Remarkable!
Learning My N-P-K's (part 2)
In my last blog, I talked about my struggle with the chemistry, the science of gardening, the bones that make it all work--not my forte. However, I decided to face it as a mature gardening adult, and learn, for starters, my N-P-Ks, those three mysterious numbers found on boxes of fertilizers and bottles of plant food. As I mentioned in my last blog (May 1), the first number listed stands for nitrogen, "N", which is essential for for healthy leaf growth. I discussed what your plants might look like if they were nitrogen deficient, and some suggestions to solve the problem.
Now we come to the second number, which informs the user of the percentage of phosphorus, "P", contained in the product inside.
I have been told (by a former science teacher, no less) that phosphorus has been used since WWI up through present day wars as a smoke tracer, an explosive, a poison and nerve gas, and has been called "the Devil's element". So why would I want this destroyer of life in my benign veggie garden? Well, it turns out that phosphorus comes in many forms and colors. It actually takes up 1 % of each one of our body weights, and is found in each cell of the human body without smoking or poisoning. It's primary use in our body, unlike in Vietnam, is the formation of bones and teeth. And just to prove to you phosphorus' versatility, it is the primary ingredient found on the head of a safety match. Believe me, this is all very confusing to a science dropout.
Phosphorus was actually discovered by a man from Munich, Germany named Hennig Brand in 1669, who found it while experimenting with urine, producing a product that actually gave off a pale green glow. He kept his method secret, as he, along with all alchemists of the day, was planning on turning his discovery into gold. He finally agreed to share his formula, and gardening has never been the same since.
But back to the question as to why any of us would want phosphorus in our gardens. Phosphorus is actually a mineral that is essential to metabolism. It is the principal element in the structure of the nucleus and cytoplasm of all tissue cells. Without it, as with nitrogen, plants do not thrive. Phosphorus is instrumental in aiding a plant convert needed nutrients into the useable building blocks that it needs to grow. If your plants are spindly, stunted in growth, have leaves that are blue/green with a purplish tinge, produce small fruit with an acid taste, or set very little fruit at all, you most likely have a phosphorus deficiency. Plants that are especially susceptible to this deficiency are carrots, lettuce, spinach, apples, currents, and gooseberries. The purplish tinge seen on the underside of tomato leaves indicates a "P" problem.
These deficiencies occur primarily in areas where there is a high level of rainfall, especially if the soil is acidic, clay, or poor chalk. Cold weather can also be a factor in the poor absorption of phosphorus.
The remedy is simple. Add organic material such as fish fertilizer as a soil drench, bone meal, colloidal phosphate or compost (food wastes are quite high in phosphorous). Also, the addition of rock phosphate is highly recommended. Edward C. Smith, author of The Vegetable Gardener's Bible, states that as phosphorus is most likely to be the nutrient missing from the soil, adding rock phosphate whenever you start a new garden is a wise move. He recommends 10 pounds per 100 square feet. Be forewarned, however. Plants that naturally adapt to low levels of available phosphate are more likely to suffer from phosphate poisoning. Aha! Back to a war reference.
Mr. Smith also makes another interesting point; do not be misled when you read numbers on the package of rock phosphate (0-4-0) that its phosphate contents is only 4%. That 4% represents the immediate available phosphorus. The product actually is about 30% phosphorus with more than 25% of it in a slow release that will become available over time.
We have one more letter to explore in our nutrient alphabet, and in my next blog I will discuss that third label number. Mmmmmm. Could "K" stand for "Kompost"?
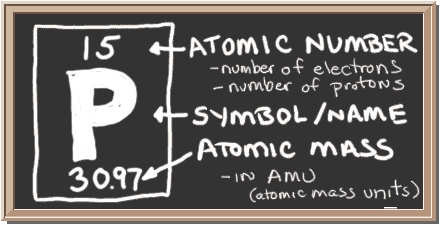
Phosphorus symbol from the elemental chart. (graphic from www.blackdiamond.org)
Thistle Be Delicious!
Almost by accident I have a multi-stemmed clump of artichokes growing at the front of my yard. This great big shrub was a transplant from an awkward space right at the edge of my driveway. I think it was a donation of a bird passing by, or some long lost endeavor from a previous homeowner. Anyway, it is thriving and continues to surprise me. I was asked the other day by one of my neighbors what kind of fertilizer I use. Pause, kind of an awkward moment, as I am sort of a lazy gardener. In the past, I have stopped by Starbucks to pick up their used coffee grounds. The soil in the front garden is clay, of course, and mine is very compacted. I figure that the coffee grounds might help loosen the soil and allow more air and moisture to reach down to the roots of the struggling plants. I also drag the grass clippings from the back yard and spread them across this bed. But beyond these 2 amendments, nothing else has contributed to the prolific offerings we have enjoyed in the last couple of years.
When I dug up the driveway volunteer, I really gave little thought to the outcome. If it made it, fine. No big investment besides a little time with my shovel and some occasional water.
In fact, at the time I considered eating artichokes hardly worth the bother. The leaves frequently were so tough, the only enjoyment was when I finally got to the heart of the choke. Maybe good-tasting, but not so healthy. I hadn't really learned to cook the chokes well and thought smothered in butter or mayonnaise was the way to go.
My mother didn't cook artichokes for us until my early teens, forget the year. It must have been one of those trendy dishes that popped up in the 60's. Not that she was a great cook, but she did like to experiment. So now that I have so many chokes, I figure I should spend a little time and give these little jewels some respect. I have found that although the leaves can bring pleasure scrapping the flesh away, the heart is where the bang is. I now steam the chokes in water with the juice of one of my Meyer lemons. I then toss them in a marinade of olive oil and balsamic vinegar, once I trim the leaves and thistle. Before serving I simmer the chokes with a good bit of the stem in a light marinara sauce. This results in a much healthier alternative to the mayo or butter versions.
Inevitably we tire of chokes and I leave a few on the shrub to let them develop the beautiful purple choke flowers. The artichoke, like many of my favorite plants, originated in the Mediterranean. It is rather a large coarse looking shrub that reaches a good 6 feet in height and width, although Sunset says 4'x 6'. The shrub grows rather quickly in the spring. One day I just notice, oh there is the artichoke shrub again. Supposedly, once I harvest the last of this first crop, I should cut the main stem an inch above the ground. New sprouts are supposed to appear and produce a second crop. No harm in trying, maybe I'll get more chokes later this summer.

The artichokes. (photo by Trisha Rose)



